MedicalDevice Manufacturing License is required to legally produce medical devices in
India. The application involves meeting strict regulatory guidelines,
submitting technical documents, and undergoing inspections. Agile Regulatory
simplifies this complex process by handling the paperwork, ensuring compliance,
and coordinating with regulatory authorities. Our expertise helps you get
licensed quickly and efficiently, allowing you to focus on manufacturing
quality devices. Contact us for smooth Medical Device Manufacturing License
registration today.
Medical Device Manufacturing License

Written by
agileregulatory
606 days ago
Related articles:
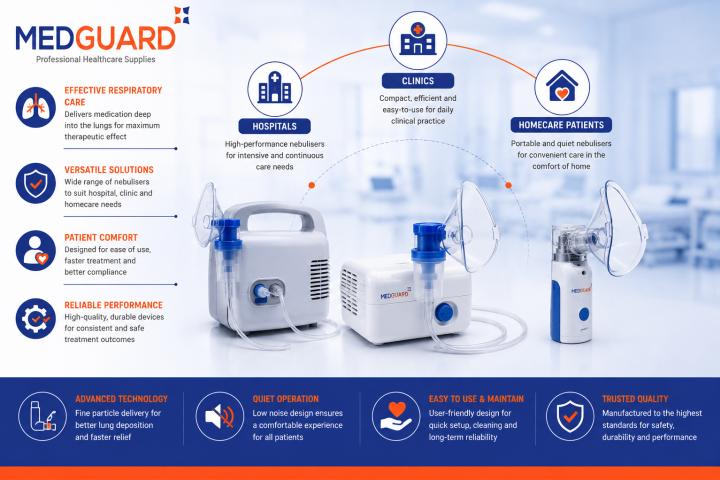
Improving Nebuliser Efficiency Through Better Infection Control Practices
Nebulisers are widely used in hospitals, clinics, nursing homes, and homecare environments to deliver respiratory medication directly into the lungs. They play an important role in treating conditions such as..
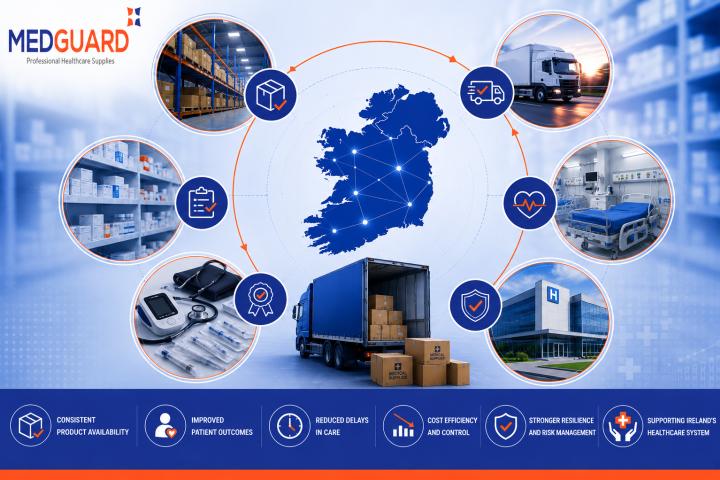
Common Challenges in Ireland’s Medical Supply Chain and How Healthcare Providers Are Responding
Ireland’s healthcare sector depends heavily on efficient medical supply chains to ensure hospitals, clinics, laboratories, and healthcare professionals have consistent access to essential medical equipment and healthcare products. From diagnostic..
Medical Equipment Rental Market Demand, Regional Insights, and Future Growth Opportunities
Medical Equipment Rental Market Growth and TrendsThe global Medical Equipment Rental Market is experiencing steady growth, driven by rising healthcare costs, increasing demand for cost-effective medical solutions, and the growing..
LINTEX Medical Scrubs Guide to Comfort, Performance and Fit
LINTEX Medical Scrubs Guide to Comfort, Performance and Fit
Build HIPAA Compliant Medical Voice Assistant: A Complete Guide
Healthcare is at a turning point where efficiency, accessibility, and patient experience are no longer optional—they are expected. Hospitals, clinics, and digital health providers are under constant pressure to reduce..
How the Right Seating Can Reduce Pain and Boost Recovery Outcomes
Recovery is a process that requires careful attention to comfort, support, and daily habits. While treatments and therapies are essential, the role of seating is often underestimated. The right seating..
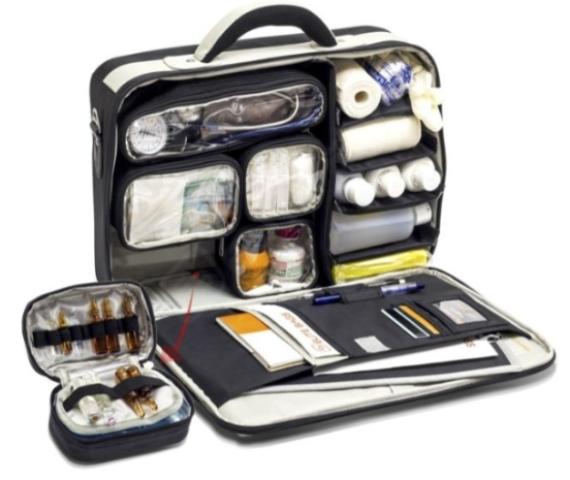
Why Every Doctor and Nurse Needs a High-Quality Medical Bag
IntroductionDoctors and nurses across Ireland work in demanding environments where efficiency, reliability, and preparedness are essential. Whether in hospitals, GP practices, care homes, or community settings, having the right equipment..
Sterile vs Non-Sterile Gloves: What’s the Difference and When to Use Them?
Medical gloves are essential for maintaining hygiene and preventing contamination in healthcare environments. One of the most important distinctions is between sterile and non-sterile gloves. Understanding their differences helps ensure..
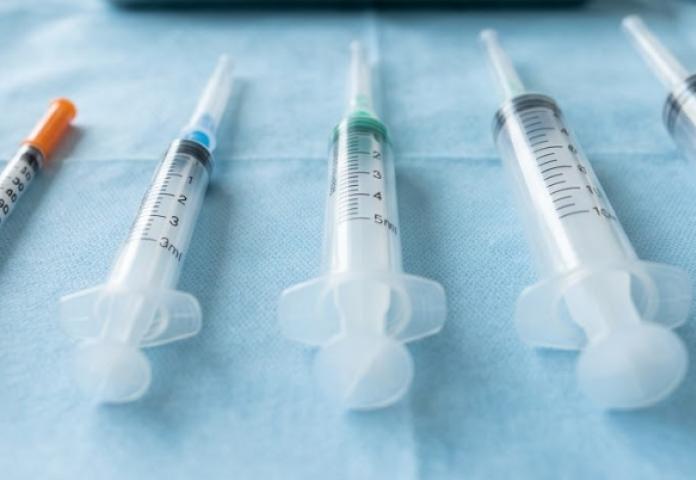
From Insulin to Safety Syringes: A Guide to Medical Syringe Types
Syringes are essential tools in modern medicine, used in hospitals, clinics, laboratories, and home care settings. They play a vital role in delivering medications, vaccines, and fluids safely and accurately...
How to Avoid CDSCO Registration Rejection (Complete Guide)
Getting approval under CDSCO registration is a crucial step for entering the medical device market in India. But many businesses face rejection—not because their product is poor, but due to..
Best UI Practices for IoT Device Control Apps
Designing the user interface for IoT device control apps is a strategic responsibility that goes far beyond aesthetics. These applications serve as the command centre for smart devices across homes,..
Understanding Device Fingerprinting: How It Tracks You and Your Privacy
Every time we go online, the using a phone, a pill, or a laptop, we leave small traces in the background. These traces can create a kind of “digital fingerprint”..
Microcurrent Device Benefits for Skin Rejuvenation and Facial Contouring
Healthy skin reflects consistent care and attention. Many people look for tools that support firmness, improve texture, and refresh tired-looking areas without adding complicated steps to their routines. A microcurrent..
Microcurrent Device Benefits for Skin Tone and Facial Definition
A microcurrent device offers a practical way to support firmer, smoother-looking skin at home. It uses low-level electrical currents that mirror the body’s natural signals. These currents interact with facial..
Beauty Device Market Analysis with Regional Insights and Future Outlook
The global Beauty Device Market is witnessing substantial growth, driven by increasing consumer focus on personal grooming, rising demand for at-home beauty solutions, and advancements in skincare technologies. According to..
How to Avoid CDSCO Registration Rejection (Complete Guide)
Getting approval under CDSCO registration is a crucial step for entering the medical device market in India. But many businesses face rejection—not because their product is poor, but due to..
Key Trends Driving Growth in 3D Printing Medical Device Software Market During 2025–2033
3D Printing Medical Device Software Industry Outlook:Straits Research has added a report titled “Global 3D Printing Medical Device Software Market Professional Report 2025-2033” to its database of research reports. The..
How to Get CDSCO Registration for Medical Devices in India (Step-by-Step Guide) Introduction
CDSCO Registration for Medical Devices in India is a mandatory process for businesses that want to manufacture, import, or sell medical devices legally. The approval is regulated under the Medical..